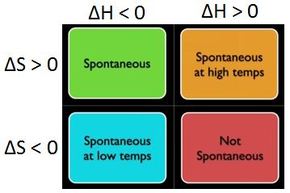
Gibbs Free Energy and Spontaneity
Introduction
In my previous couple of blog posts, I talked about a thermodynamic state function called enthalpy, and how it is used by scientists and engineers. This included covering a principle called Hess’s Law, which has led to the tabulation of enthalpy values for certain reactions under a set of standardized conditions, such that the idea could be generalized to make thermodynamic predictions about a huge variety of processes.
Those posts laid the groundwork for the following topic. (more…)